Building 12, No. 2317 Shengang Road, Songjang, Shanghai, China. +86-150 2197 1920 [email protected]
Austenite:

The solid solution of carbon and alloying elements dissolved in γ-Fe still retains the face-centered cubic lattice characteristics of γ-Fe. This structure generally exists at high temperatures; austenite begins to decompose at 200-300°C. As the heating temperature increases, the grains gradually grow larger. At a given temperature, the longer the holding time, the coarser the austenite grains. The grain boundaries are relatively straight, forming regular polygons; it is non-magnetic, has good plasticity, low strength, and certain toughness; in quenched steel, residual austenite is distributed in the gaps between martensite needles.
Supercooled austenite:
Austenite that exists below the A1 temperature and is unstable and about to undergo transformation.
Ferrite:
A solid solution of carbon and alloying elements dissolved in α-Fe, possessing a body-centered cubic lattice and extremely low carbon solubility; Characteristics: It exhibits good toughness and plasticity; it presents a bright, polygonal grain structure; it exists at high temperatures above 1400℃, hence it is called high-temperature ferrite or δ solid solution, denoted by δ; in hypoeutectoid steel, slowly cooled ferrite appears as blocky grains with relatively smooth grain boundaries. When the carbon content approaches the eutectoid composition (0.77% carbon content), ferrite precipitates along the grain boundaries. (Eutectoid: A phase transformation in which two or more new phases precipitate together from the parent phase.)
Martensite:

A supersaturated solid solution of carbon dissolved in α-Fe, with a body-centered tetragonal structure; common martensite morphologies include lath and plate-like structures; the morphology of martensite mainly depends on the formation temperature, which in turn depends on the content of carbon and alloying elements in the austenite; for carbon steel, as the carbon content increases, the amount of lath martensite relatively decreases, and the amount of plate-like martensite relatively increases; characteristics: high strength and high hardness; formed by rapid cooling (quenching) of austenite, it is not an equilibrium structure and easily decomposes when heated to 80–200°C;
Lath martensite:
Formed in low- and medium-carbon steels and stainless steels, it consists of bundles of laths arranged in parallel. The spatial shape is flattened and elongated, and one austenite grain can transform into several lath bundles (usually 3 to 5).
Lath martensite (acicular martensite):
This is commonly found in high and medium carbon steels and high-nickel Fe-Ni alloys. When the largest martensite laths are too small to be resolved by an optical microscope, it is called cryptocrystalline martensite. The martensite obtained from normal quenching in production is generally cryptocrystalline martensite.
Tempered Martensite:
This microstructure is formed by low-temperature tempering (150–250°C) and consists of martensite with a lower degree of supersaturation and very fine carbides. Martensite decomposition occurs between 80 and 200°C. When the steel is heated to approximately 80°C, the atomic activity within it increases, and the supersaturated carbon in the martensite begins to precipitate gradually in the form of carbides. The degree of carbon supersaturation in the martensite continuously decreases, thus forming a mixed structure of martensite with a lower degree of supersaturation and very fine carbides.
Cementite:
A compound of carbon and iron, Fe3C; Characteristics: Contains 6.67% carbon, has a complex orthorhombic crystal structure; very hard, extremely brittle, with almost zero toughness and plasticity;
Pearlite:
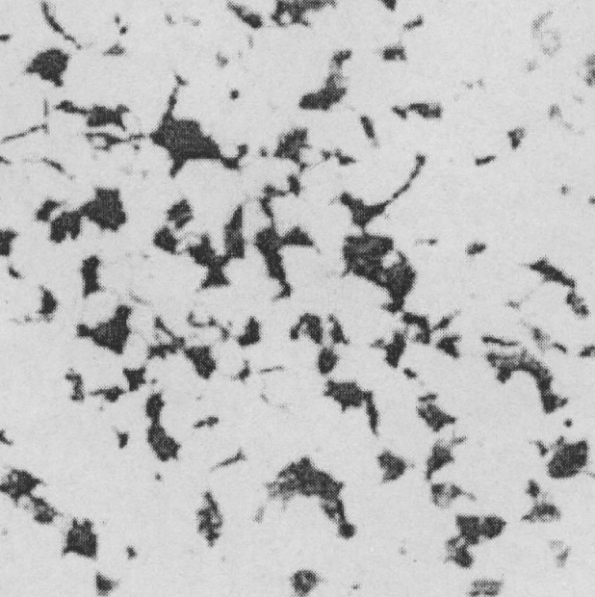
A mechanical mixture of ferrite and cementite in alternating lamellar layers, formed by the eutectoid reaction in iron-carbon alloys; Characteristics: Exhibits a pearly luster; its mechanical properties are between those of ferrite and cementite, with high strength, moderate hardness, and good plasticity and toughness;
Lamellar Pearlite:
A mixture of ferrite and cementite formed by alternating overlapping thin layers; Based on the size of the lamellar spacing, it can be divided into: Pearlite (lamellar spacing 450–150 nm, formation temperature range A1–650℃, clearly distinguishable under an optical microscope), Sorbite (lamellar spacing 150–80 nm, formation temperature range 650–600℃, only distinguishable under a high-magnification optical microscope), and Troostite (lamellar spacing 80–30 nm, formation temperature range 600–550℃, only distinguishable with an electron microscope);
Granular Pearlite:
A mixture in which cementite exists in granular form within a ferrite matrix; Granular pearlite is generally obtained through spheroidizing annealing; (Spheroidizing annealing: Annealing performed to spheroidize the carbides in steel);
Upper Bainite:
A mixture formed from supersaturated acicular ferrite and cementite when the temperature drops to the range of 550–350℃, with cementite between the ferrite needles; Characteristics: Feather-like appearance, brittle, high hardness; basically identifiable under a 500x optical microscope. Lower Bainite
A mixture formed from supersaturated acicular ferrite and cementite when the temperature drops to the range of 350–230℃, but the cementite is within the ferrite needles; Characteristics: appears as black needle-like or bamboo leaf-like structures;
Granular Bainite:
A mixture composed of relatively coarse blocky ferrite and carbon-rich austenite;
Carbide-Free Bainite:
A microstructure composed of a single phase of lath-like ferrite, also known as ferritic bainite; Characteristics: Carbide-free bainite generally appears in low-carbon steel;
Widmanstätten Structure:
A multiphase microstructure in steel where the proeutectoid phase exists in a needle-like or plate-like form mixed with lamellar pearlite, occurring when the austenite grains are relatively coarse and the cooling rate is appropriate. Characteristics: Coarse grains, with plate-like, feathery, or triangular shapes.
 Hot News
Hot News